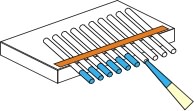
Equilibrium Dialysis with acrylamide plugs.
|
|
|
|
|
|
|
|
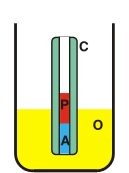 | |
| C P O A |
- Capillary - Protein - Outer solution - Acrylamide plug |
Zeppezauer et al. [Zeppezauer, 1968] described the use of polyacrylamide, polymerized in capillaries, as a dialysis membrane for the crystallization of proteins. In this procedure capillaries are dipped in an acrylamide solution to obtain a liquid column of about 1 cm at the end of the capillaries. After polymerization the capillaries are filled with buffer and placed for 2-3 days in the start buffer to allow residual traces of acrylamide monomers and polymerization catalysts to diffuse out of the gel. Crystallization experiments are started by withdrawing the buffer with a thin tubing and the injecting the protein solution into the cell. The upper end of the capillary is closed with paraffin. The capillary is placed vertically into the outer solution standing on Pyrex-wool. If crystallization occurs only at high salt or solvent concentration, the polyacrylamide plug can shrink excessively and detach from the inside wall of the capillary. In such cases, the polyacrylamide plug should be prepared from a monomer solution containing the buffer and about half the concentration of salt required for crystallization.
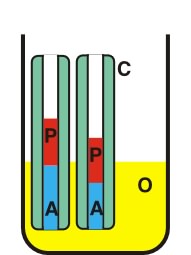 | |
| C P O A |
- Capillary - Protein - Outer solution - Acrylamide plug |